【Brain Health Ingredient Series 4】 Citicoline (CDPC): The “Phospholipid Precursor” for Neural Repair
Apr. 17, 2026
1. What is Citicoline (CDPC)?
Cytidine-5'-diphosphocholine, also known as Citicoline (CDPC), is a phospholipid precursor that can be synthesized endogenously in the human body. It is an essential component of neuronal cell membranes and consists of cytidine and choline. Citicoline serves as both a precursor for phosphatidylcholine (the main phospholipid of neuronal membranes) and for the neurotransmitter acetylcholine, making it a 'dual nutrient for neural repair.' Small amounts of Citicoline are found in foods such as animal liver, fish, and eggs, but supplementation is often required to achieve effective levels.
2. Basic Characteristics
Citicoline is a white, odorless powder with high solubility and a bioavailability of up to 90%. After oral intake, it rapidly hydrolyzes into cytidine and choline, both of which can cross the blood–brain barrier and reach neurons directly [1]. Within the brain, cytidine can convert into uridine, which works synergistically with choline to synthesize phosphatidylcholine, facilitating the repair of damaged neuronal membranes [5]. Additionally, its metabolites enhance neurotransmitter synthesis [7], providing a dual role of 'repair and regulation' with minimal metabolic burden. Common production methods include microbial fermentation, organic synthesis, and enzymatic catalysis.
3. Functional Effects: From Neural Repair to Cognitive Enhancement
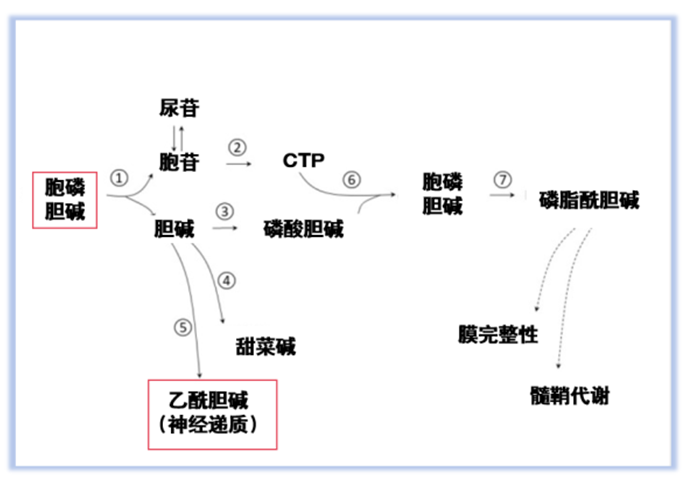
Image source: Świątkiewicz M, et al. Aging Dis. 2023 Aug 1;14(4):1184-1195.
• Repairing neuronal membranes: Citicoline promotes phospholipid synthesis [16] and strengthens membrane stability in response to damage or aging [13], making it beneficial for individuals experiencing brain fatigue or memory decline.
• Enhancing cognition and memory: By regulating the metabolism of acetylcholine [3,11], dopamine, and other neurotransmitters, Citicoline improves neural signal transmission, attention, and memory recall.
• Supporting cerebrovascular health and reducing fatigue: Citicoline contributes to mitochondrial respiration [2] and ATP synthesis, supplying sufficient energy for neuronal activity, increasing cerebral blood flow, enhancing oxygen delivery, and reducing lactic acid buildup [14] to relieve mental fatigue.
4. Regulatory Status
• European Union: In July 2014, the EU issued Decision 2014/423/EU approving Citicoline as a novel food ingredient for dietary supplements targeting middle-aged and elderly populations. The maximum allowable intake is 500 mg/day for supplements and 250 mg per serving for foods for special nutrition; not suitable for children.
• China: In 2014, the National Medical Products Administration (NMPA) issued a notice revising the instructions for Citicoline sodium injections (Document No. 2014-201). Currently, it is primarily regulated as a pharmaceutical ingredient.
• Australia & New Zealand: In February 2024, Food Standards Australia New Zealand (FSANZ) approved Citicoline as a nutrient for use in caffeine-containing formulated beverages (Application A1290).
5. Clinical Evidence: Data-Supported Neuroprotective Efficacy
• The Citicholinage Study, a retrospective, multicenter case-control study conducted across seven dementia centers in Italy, enrolled 448 Alzheimer’s disease (AD) patients aged 65 and above. Results showed that patients treated with Citicoline combined with cholinesterase inhibitors experienced statistically significant improvements in Mini-Mental State Examination (MMSE) scores, suggesting delayed AD progression.
• In a randomized controlled trial by Nakazaki et al. [12], participants who took 500 mg/day of Citicoline for 12 weeks showed marked improvements in episodic and overall memory scores compared with the placebo group, indicating beneficial effects on memory in middle-aged and older adults.
• A 2022 meta-analysis reviewing multiple trials [10] on Citicoline in acute and subacute stroke treatment reported significant cognitive recovery benefits—particularly in memory and attention—along with a trend toward reduced post-stroke disability.
6. Safety Profile
• Acute toxicity tests showed that rats administered 2000 mg/kg of Citicoline exhibited no notable adverse effects such as nausea or dizziness.
• Long-term safety studies (1200 mg daily for 12 months) revealed no abnormalities in liver, kidney, or hematological parameters, and no drug dependence. Mild adverse events such as digestive discomfort or headaches were occasionally reported.
• Citicoline should be avoided by children, pregnant and lactating women, and those allergic to choline compounds.
6. Recommended Dosage
• For daily brain health maintenance (healthy adults): 250–1000 mg/day, may be combined with DHA for synergistic neuronal membrane repair.
• For targeted improvement (mild cognitive impairment or mental fatigue): 1000–2000 mg/day, recommended for 8–12 weeks of continuous supplementation for stable results.
8. Precautions
• Prolonged use of Citicoline may cause mild gastrointestinal discomfort, such as nausea, vomiting, or diarrhea. These symptoms are likely related to mild irritation of the gastrointestinal tract.
• Individuals taking anticoagulants (e.g., warfarin), antihypertensive agents, or antiepileptic drugs should consult a physician before using Citicoline to avoid potential interactions. For long-term users, regular follow-up is recommended to monitor cognitive performance as well as liver and kidney function to ensure both efficacy and safety.
• In rare cases, Citicoline may cause anxiety or agitation, especially during prolonged treatment. Therefore, individuals with a history of anxiety, depression, or other emotional disorders should use it with caution.
• Citicoline is contraindicated in pregnant or breastfeeding women due to insufficient safety data. It is also not recommended for minors because of a lack of clinical evidence. Elderly individuals should start with a low dose and undergo regular liver and kidney function monitoring. It is contraindicated in patients allergic to Citicoline, and should be used cautiously during the acute phase of hemorrhagic diseases.
• Citicoline is not a substitute for medical treatment. For patients diagnosed with cognitive disorders such as Alzheimer’s disease, Citicoline should be used only as an adjunctive supplement under medical supervision.
References:
[1]李红霞,王晓丽,来韵祺. 胞磷胆碱钠联合认知功能训练治疗脑卒中后认知功能障碍的效果观察[J].全科医学临床与教育,2025,23(09):816-819.DOI:10.13558/j.cnki.issn1672-3686.2025.009.009.
[2]Aziz A N ,Algeda R F ,Shedid M S . The impact of citicoline on brain mitochondrial dysfunction induced in rats after head irradiation[J].Scientific Reports,2025,15(1):25658-25658.DOI:10.1038/S41598-025-11098-4.
[3]Bernhard W ,Shunova A ,Mainka G U , et al. Choline in Pediatric Nutrition: Assessing Formula, Fortifiers and Supplements Across Age Groups and Clinical Indications[J].Nutrients,2025,17(10):1632-1632.DOI:10.3390/NU17101632.
[4]于克文,高宇,孙翠梅. 神经保护药物治疗脑梗死效果分析[J].中国城乡企业卫生,2025,40(04):113-115.DOI:10.16286/j.1003-5052.2025.04.042.
[5]Hadzibegovic S ,Nicole O ,Andelkovic V , et al. Examining the effects of extremely low-frequency magnetic fields on cognitive functions and functional brain markers in aged mice[J].Scientific Reports,2025,15(1):8365-8365.DOI:10.1038/S41598-025-93230-Y.
[6]高昶,刘燕,聂琼,等. 胞磷胆碱治疗卒中后认知障碍患者的临床研究[J].中国临床药理学杂志,2024,40(19):2801-2805.DOI:10.13699/j.cnki.1001-6821.2024.19.009.
[7]Nakazaki E ,Lili L ,Zhang B , et al. Investigating Specific Molecular and Functional Impacts of Citicoline on Brain Health[J].Current Developments in Nutrition,2024,8(S2):103222-103222.DOI:10.1016/J.CDNUT.2024.103222.
[8]Gareri P ,Cotroneo M A ,Montella R , et al. Citicoline: A Cholinergic Precursor with a Pivotal Role in Dementia and Alzheimer's Disease.[J].Journal of Alzheimer's disease : JAD,2024,100(2):725-733.DOI:10.3233/JAD-240497.
[9]Mehdi M ,Bahram A ,Mehdi S , et al. Citicoline on the Barthel Index: Severe and moderate brain injury.[J].Indian journal of pharmacology,2023,55(4):223-228.DOI:10.4103/IJP.IJP_570_21.
[10]Moein K ,Fatemeh M ,Maede A , et al. Citicoline for traumatic brain injuries: A systematic review and implications for future research[J].Archives of Trauma Research,2022,11(3):140-149.DOI:10.4103/ATR.ATR_51_22.
[11]P. O M ,V. N C ,G. G S . Effects of Citicoline on Structural/Functional Consequences of Focal Ischemia of the Rat Brain[J].Neurophysiology,2022,53(2):78-87.DOI:10.1007/S11062-022-09918-8.
[12]Eri N ,Eunice M ,Kristen S , et al. Citicoline and Memory Function in Healthy Older Adults: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial.[J].The Journal of nutrition,2021,151(8):2153-2160.DOI:10.1093/JN/NXAB119.
[13]杜文军,李青,刘丽爽. 胞磷胆碱胶囊联合神经肌肉刺激治疗脑损伤疗效及对认知功能与功能独立性康复影响[J].临床军医杂志,2019,47(09):952-953+956.DOI:10.16680/j.1671-3826.2019.09.23.
[14]Marek H ,Sophie M ,Marie-Aude B L , et al. Clinical, biometric and structural evaluation of the long-term effects of a topical treatment with ascorbic acid and madecassoside in photoaged human skin.[J].Experimental dermatology,2008,17(11):946-52.DOI:10.1111/j.1600-0625.2008.00732.x.
[15]朱正禹,毕建忠,来超,等. 不同剂量胞磷胆碱治疗急性脑梗死的疗效和副作用[J].河南实用神经疾病杂志,2003,(04):41.DOI:CNKI:SUN:HNSJ.0.2003-04-034.
[16]Le\'on-Carri\'on J ,Dominguez-Rold\'an M J ,Murillo-Cabezas F , et al. The role of citicholine in neuropsychological training aftertraumatic brain injury[J].Neurorehabilitation,2000,14(1):33-40.
Latest Blogs
-
May. 07, 2026
【Brain Health Ingredient Series 7】Taurine – The “Natural Amino Acid Guardian” for Brain Protection
-
Apr. 27, 2026
-
Apr. 22, 2026
Exhibition Recap | L&P Foods Drives Quality & Efficiency at 2026 China Feed Industry Expo
-
Apr. 20, 2026
-
Apr. 17, 2026
【Brain Health Ingredient Series 4】 Citicoline (CDPC): The “Phospholipid Precursor” for Neural Repair




