【Brain Health Ingredient Series 6】 L-α-Glycerylphosphorylcholine (GPC): The “Choline Donor” That Activates the Brain
Apr. 27, 2026
- What is Glycerylphosphorylcholine (GPC)?
L-α-Glycerylphosphorylcholine (L-alpha-GPC, commonly referred to as L-α-GPC or GPC) is a naturally occurring phospholipid compound. It is an essential component of human cell membranes and is widely distributed in tissues such as the brain and liver, with particularly high concentrations in the brain. It can also be obtained from dietary sources including egg yolk, milk, and soybeans.
As an active form of choline, GPC can directly supply choline to the brain without the need for complex metabolic conversion [1], and is therefore often described as a “rapid choline replenishment system in the brain.”
Functionally, GPC plays a critical role as a precursor for the synthesis of acetylcholine—a neurotransmitter closely associated with memory and cognition—as well as phosphatidylcholine, a key structural component of cell membranes. In addition, it participates in lipid metabolism [11], neuroprotection, and cellular signal transduction, thereby providing essential support for maintaining normal physiological functions.
- Basic Properties
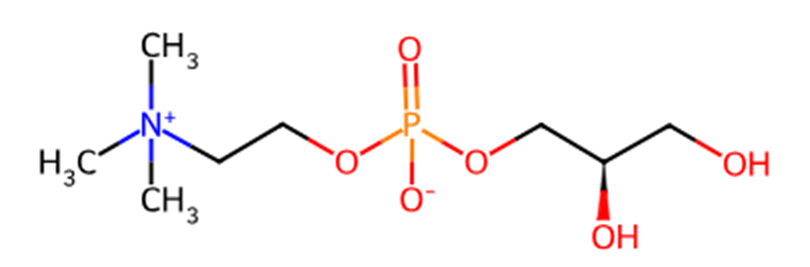
Image source: ChemSpider
GPC is a white, odorless powder with a slightly sweet taste. It is fully miscible with water, soluble in methanol and ethanol, and insoluble in chloroform and ether.
As a small-molecule phospholipid compound, it can be directly absorbed through the intestinal tract after oral administration, with a significantly higher absorption rate than conventional choline sources. It is also able to readily cross the blood-brain barrier. After entering the brain, approximately 90% of GPC is converted into choline within one hour, rapidly meeting the brain’s demand for choline.
GPC exhibits both excellent water solubility and lipid solubility, making it suitable for various formulation types. In addition, it is chemically stable and resistant to oxidation, which supports its suitability for long-term supplementation. Currently, it is mainly produced through chemical synthesis, biosynthesis, and aqueous enzymatic methods [8].
3. Functional Benefits: Multifaceted Effects from Cognitive Enhancement to Neuroprotection
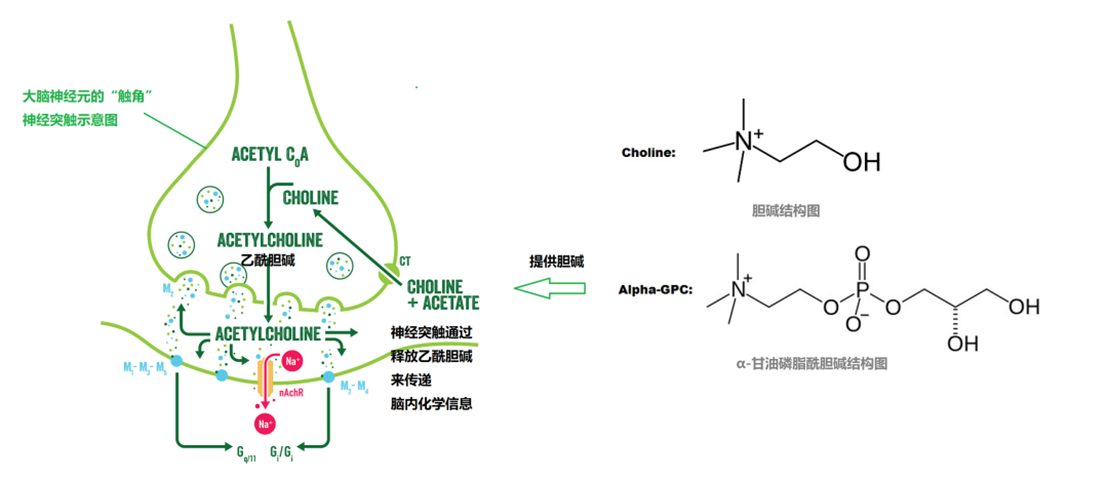
Image source: Supplement Frontiers
Enhances brain function and cellular vitality:
GPC is metabolized in the brain into glycerol-3-phosphate and choline, serving as an efficient reservoir for acetylcholine [7]. By promoting the synthesis and release of acetylcholine, it helps enhance memory and improve cognitive performance.
Neuroprotection and cognitive improvement:
By supplying choline to the brain and promoting acetylcholine synthesis [3], GPC enhances the efficiency of neural signal transmission [10]. It helps improve issues such as poor concentration and difficulty in memory recall, making it particularly suitable for elderly individuals with cognitive decline and those engaged in high-intensity mental work.
Antioxidant effects and protection against neuronal aging:
GPC promotes brain glucose metabolism, providing energy for neural activity and alleviating mental fatigue [5]. It also helps regulate neurotransmitter balance and supports mood improvement [6]. As a precursor of membrane phospholipids, GPC can repair damaged neuronal cell membranes, enhance cellular stability, and reduce oxidative stress-induced neuronal damage [9], thereby lowering the risk of neurodegenerative diseases such as Alzheimer’s disease.
4. Regulatory Status: A Globally Recognized Safe Nutritional Ingredient
- United States:
In 2012, GPC (synthetic) was granted GRAS status by the FDA (GRN No. 419). It is permitted for use in conventional foods, including beverages, beverage bases, and snack products such as chocolate, candy, and chewing gum.
- European Union:
According to Regulation (EC) No. 2017/2470, GPC is classified as an unauthorized novel food and is not yet permitted for use in food products. Market entry requires prior authorization.
Currently, a novel food application for soybean-derived GPC (submitted by Chemi S.p.A for use in food supplements) has been accepted by the European Commission, and its safety is under evaluation by EFSA (EFSA-Q-2023-00492).
In Switzerland, the Federal Food Safety and Veterinary Office (FSVO) has approved GPC as a novel food.
- China:
In 2024, the National Health Commission approved GPC (synthetic) as a New Food Ingredient (Announcement No. 2, 2024). It is permitted for use in food products, with a recommended intake of ≤ 600 mg/day (dry basis).
- Canada:
In 2023, GPC was approved as a Natural Health Product (NHP) and is listed in the Natural Health Products Database, categorized under cognitive health products.
- Australia:
GPC is classified as a complementary medicine.
- Japan:
In 2009, GPC was included in the list of ingredients that are not classified as pharmaceuticals unless specific efficacy claims are made, and it has since been recognized as a novel food ingredient, widely used in the development of functional foods.
5. Clinical Evidence: Data-Validated Cognitive Benefits
- A 2003 study published in Archives of Gerontology and Geriatrics [12] involving 60 patients with mild cognitive impairment (average age 72) showed that daily supplementation with 300 mg of GPC for 6 months resulted in a 28% improvement in memory scale scores, with the most notable enhancements observed in the recall of recent events and recognition of familiar objects.
- A 2015 prospective case-control study [13] including 34 participants aged 65–85 without a history of dementia found that those in the experimental group who received 800 mg of GPC daily for 11 months demonstrated superior language comprehension ability compared to the control group, as assessed by pure-tone audiometry and bilateral word recognition tests.
- A 2023 meta-analysis published in Journal of Alzheimer’s Disease [4], which included 8 studies involving a total of 861 patients from multiple countries with study durations ranging from 90 to 720 days, demonstrated that Alzheimer’s patients receiving GPC showed an average increase of 1.72 points in MMSE scores and a reduction of 5.76 points in ADAS-Cog scores, confirming that GPC has a clear and significant beneficial effect on cognitive dysfunction.
6. Safety Profile: Excellent Tolerability for Long-Term Supplementation
- Acute Toxicity:
Animal studies have shown that even at high oral doses (with single-dose administration in rats far exceeding the recommended human intake by several times), no significant toxic symptoms or mortality were observed. This indicates a high acute safety threshold, with no risk of acute toxicity under normal oral supplementation.
- Long-Term Safety:
A long-term study (1,200 mg/day for 12 months) demonstrated that subjects showed no abnormalities in liver or kidney function, hematological parameters, or coagulation function. No dependency or withdrawal (rebound) effects were observed.
7. Recommended Dosage
- China: Recommended intake ≤ 600 mg/day (dry basis), as specified by the National Health Commission
- Canada: Recommended intake ≤ 1200 mg/day
- Targeted Cognitive Support (e.g., mild cognitive impairment, mental fatigue):
800–1200 mg/day, divided into 2–3 doses
Recommended duration: 6–8 weeks
After stabilization of cognitive condition, the dosage may be reduced to 400–600 mg/day as a maintenance level
8. Precautions
GPC is hygroscopic and should be stored in sealed containers; prolonged exposure to air should be avoided.
- Not recommended for infants, pregnant women, or breastfeeding women.
- Individuals with severe liver or kidney impairment should adjust the dosage under medical supervision to avoid metabolic burden.
- Not a substitute for medication — for diagnosed cognitive disorders (such as Alzheimer’s disease), GPC should be used only as a supportive nutritional supplement alongside prescribed medical treatment.
References
[1]Ji Y ,Li X ,Bian Y , et al. A novel chiral HPLC method development and validation for sensitive detection of isomeric impurities in L-α-glycerylphosphorylcholine (L-α-GPC) from distinct sources: chemical synthesis versus bio-derivative[J].Microchemical Journal,2025,217115036-115036.DOI:10.1016/J.MICROC.2025.115036.
[2]Cao J ,Su E . Unlocking the potential of l-α-glycerylphosphorylcholine in the food industry: From safety approvals to market prospects.[J].Comprehensive reviews in food science and food safety,2025,24(2):e70117.DOI:10.1111/1541-4337.70117.
[3]Soyogu Y ,Naoki K ,Wei W , et al. Effects of egg yolk choline intake on cognitive functions and plasma choline levels in healthy middle-aged and older Japanese: a randomized double-blinded placebo-controlled parallel-group study[J].Lipids in Health and Disease,2023,22(1):75-75.DOI:10.1186/S12944-023-01844-W.
[4]Gamo G S ,Enea T ,Francesco A . Activity of Choline Alphoscerate on Adult-Onset Cognitive Dysfunctions: A Systematic Review and Meta-Analysis.[J].Journal of Alzheimer's disease : JAD,2023,92(1):59-70.DOI:10.3233/JAD-221189.
[5]Anna C ,Maria A F ,Valentino M , et al. Association Between the Cholinesterase Inhibitor Donepezil and the Cholinergic Precursor Choline Alphoscerate in the Treatment of Depression in Patients with Alzheimer’s Disease[J].Journal of Alzheimer's Disease Reports,2022,6(1):235-243.DOI:10.3233/ADR-200269.
[6]Gyeongsil L ,Seulggie C ,Jooyoung C , et al. Association of L-α Glycerylphosphorylcholine With Subsequent Stroke Risk After 10 Years.[J].JAMA network open,2021,4(11):e2136008-e2136008.DOI:10.1001/JAMANETWORKOPEN.2021.36008.
[7]Gina N ,Hyun S K ,Hyun S J , et al. Supplementary Effect of Choline Alfoscerate on Speech Recognition in Patients With Age-Related Hearing Loss: A Prospective Study in 34 Patients (57 Ears)[J].Frontiers in Aging Neuroscience,2021,13684519-684519.DOI:10.3389/FNAGI.2021.684519.
[8]Jeongeun K ,Yejin S ,Jeong S L , et al. Enzymatic preparation of food-grade l-α-glycerylphosphorylcholine from soy phosphatidylcholine or fractionated soy lecithin.[J].Biotechnology progress,2020,36(1):e2910.DOI:10.1002/btpr.2910.
[9]Lee H S ,Choi Y B ,Kim H J , et al. Late treatment with choline alfoscerate (l-alpha glycerylphosphorylcholine, α-GPC) increases hippocampal neurogenesis and provides protection against seizure-induced neuronal death and cognitive impairment[J].Brain Research,2017,1654(Pt A):66-76.DOI:10.1016/j.brainres.2016.10.011.
[10]Carotenuto A ,Rea R ,Traini E , et al. The Effect of the Association between Donepezil and Choline Alphoscerate on Behavioral Disturbances in Alzheimer’s Disease: Interim Results of the ASCOMALVA Trial[J].Journal of Alzheimer's Disease,2016,56(2):805-815.DOI:10.3233/JAD-160675.
[11]Kawamura T ,Okubo T ,Sato K , et al. Glycerophosphocholine enhances growth hormone secretion and fat oxidation in young adults[J].Nutrition,2012,28(11-12):1122-1126.DOI:10.1016/j.nut.2012.02.011.
[12]F A ,K S T . Pathways of acetylcholine synthesis, transport and release as targets for treatment of adult-onset cognitive dysfunction.[J].Current medicinal chemistry,2008,15(5):488-98.
[13]Sagaro GG, Traini E.Amenta FActivity of Choline Alphoscerate on Adult-Onset Cognitive Dysfunctions: A Systematic Review and Meta-Analysis.J Alzheimers Dis.2023;92(1):59-70.
Latest Blogs
-
May. 07, 2026
【Brain Health Ingredient Series 7】Taurine – The “Natural Amino Acid Guardian” for Brain Protection
-
Apr. 27, 2026
-
Apr. 22, 2026
Exhibition Recap | L&P Foods Drives Quality & Efficiency at 2026 China Feed Industry Expo
-
Apr. 20, 2026
-
Apr. 17, 2026
【Brain Health Ingredient Series 4】 Citicoline (CDPC): The “Phospholipid Precursor” for Neural Repair




