Canada Updates the List of Permitted Supplemental Ingredients — Phosphatidylserine (Soy) Approved as a Food Supplement Ingredient
Apr. 30, 2024

Image source: https://www.canada.ca/en.html
On April 18, 2024, Health Canada issued document M-SIS-24-01, revising the List of Permitted Supplemental Ingredients and approving phosphatidylserine (soy) for use as a food supplemental ingredient [1].
The Food Directorate of Health Canada completed a safety assessment of phosphatidylserine. The assessment concluded that information related to chemistry, nutrition, microbiology, toxicology, and allergenicity supports the safety of phosphatidylserine (soy) for use in supplemented foods.
Phosphatidylserine is a glycerophospholipid that occurs naturally in a variety of common foods. It is composed of the amino acid serine, a phosphate group, two fatty acids, and glycerol. When phosphatidylserine is produced from soy lecithin, the fatty acids constituting phosphatidylserine are primarily linoleic acid and palmitic acid.
Clinical studies have shown that phosphatidylserine (soy) is well tolerated and is not associated with reports of allergic reactions. Furthermore, when used as a supplemental ingredient under the conditions of use outlined in Part IV of the U.S. Food Codex, phosphatidylserine (soy) does not raise any toxicological or nutritional concerns and is therefore included on the List of Permitted Supplemental Ingredients [2].
Health Canada concluded that the available safety data for phosphatidylserine derived from food-grade soy lecithin support its safe use in supplemented foods, provided that the maximum daily intake does not exceed 300 mg, the amount per serving is limited to 300 mg, and that certain additional compositional and labeling requirements are met.
Required Warning Statements on Labels
(a) All products containing phosphatidylserine (soy) must include the following warnings:
· “For adults only (18 years of age or older).”
· “Not recommended for use by pregnant or breastfeeding women.”
· “Do not [eat/drink] any other supplemented foods or supplements containing the same supplemental ingredient/ (specific ingredient name) on the same day.”
(b) If the declared amount of phosphatidylserine (soy) on the label exceeds 60 mg per serving, the following additional warning statement is required:
· “Do not [eat/drink] more than X servings per day,”
where X is the number of servings that provides a total daily intake of phosphatidylserine (soy) not exceeding 300 mg, based on the labeled amount per serving.
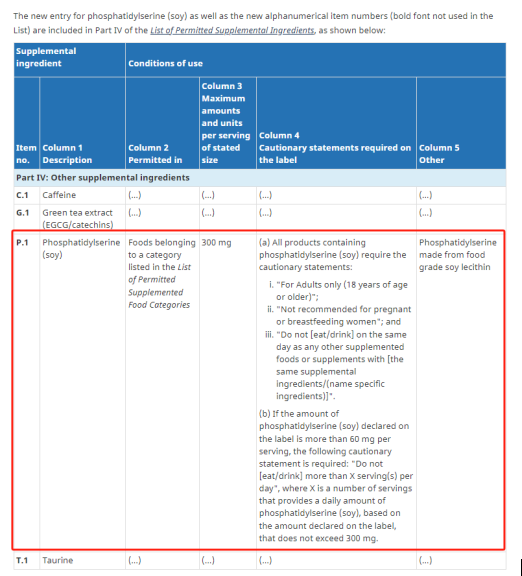
Image source: https://www.canada.ca/en.html
About L&P Food Ingredient Co., Ltd
L&P Food Ingredient Co., Ltd (L&P Foods) is a professional manufacturer specializing in food additives and functional food ingredients. The company holds comprehensive production qualifications, including valid manufacturing licenses and a full range of internationally recognized management system certifications, including Quality Management (ISO 9001), Food Safety Management (ISO 22000), Religious Certifications (KOSHER and HALAL), Occupational Health and Safety (ISO 45001), Environmental Management (ISO 14001), as well as the Global Standard for Food Safety (BRC). With consistent and reliable product quality, a well-established after-sales service system, and the capability to provide customized solutions tailored to customer needs, L&P Foods has become a trusted and preferred supplier for downstream manufacturers.
References:
[1] https://www.canada.ca/en.html
[2]Veronika V ,Tzafra C ,Yael R , et al. Phosphatidylserine containing omega-3 fatty acids may improve memory abilities in non-demented elderly with memory complaints: a double-blind placebo-controlled trial.[J].Dementia and geriatric cognitive disorders,2010,29(5): 467-74.DOI:10.1159/000310330.
Latest Blogs
-
May. 07, 2026
【Brain Health Ingredient Series 7】Taurine – The “Natural Amino Acid Guardian” for Brain Protection
-
Apr. 27, 2026
-
Apr. 22, 2026
Exhibition Recap | L&P Foods Drives Quality & Efficiency at 2026 China Feed Industry Expo
-
Apr. 20, 2026
-
Apr. 17, 2026
【Brain Health Ingredient Series 4】 Citicoline (CDPC): The “Phospholipid Precursor” for Neural Repair




