How Is the Safety of BHA Recognized as a Food Additive?
Jun. 14, 2022
June 7 marked the Fifth World Food Safety Day, with this year’s theme being “Safer Food, Better Health”. Historically, many food safety incidents have been associated with food additives or illegal substances. As a result, modern consumers tend to scrutinize ingredient lists carefully and often prefer foods with fewer or no additives.
As a food additive, butylated hydroxyanisole (BHA) has a history of use spanning more than 70 years. It is primarily applied as an antioxidant to prevent the oxidation of fats and oils. Over time, its applications have continued to expand, and it has gained regulatory acceptance in an increasing number of countries.
In an era of heightened consumer awareness around food safety, an important question remains: how safe is BHA as a food additive, and how should it be properly understood and evaluated?
What Is BHA?
Butylated hydroxyanisole (BHA), also known as tert-butyl-4-hydroxyanisole, is a mixture of two isomers, 3-BHA and 2-BHA. BHA is a synthetic antioxidant that effectively prevents the oxidative rancidity of fats and oils.
Due to its strong antioxidant performance, BHA is widely used in food and food packaging, as well as in the pharmaceutical, feed, and cosmetics industries.
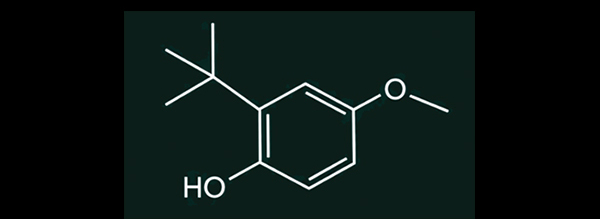
Structural Formulas of BHA Isomers
3-BHA (left) and 2-BHA (right)
Since 1947, butylated hydroxyanisole (BHA) has been added to edible fats as an antioxidant. It plays an important role in food preservation by preventing the oxidative rancidity of fats and oils and protecting oil-soluble vitamins from degradation.
In 1984, the International Life Sciences Institute (ILSI) reported that approximately 50 countries permitted the use of BHA as a food additive. Today, BHA remains present in a wide range of food products, with maximum permitted levels (MPLs) varying by food category and region, typically ranging from 2 ppm to 5,000 ppm [3–6].
In addition to its direct use as a food additive, BHA is also incorporated into food packaging materials, where it protects packaged foods through the migration or volatilization of the antioxidant, thereby enhancing oxidative stability.
Toxicological Studies of BHA
Multiple authoritative bodies, including the European Food Safety Authority (EFSA), the Joint FAO/WHO Expert Committee on Food Additives (JECFA), and Health Canada, have conducted rigorous evaluations of extensive data on the genotoxicity, subchronic toxicity, and carcinogenicity of butylated hydroxyanisole (BHA). These assessments consistently conclude that BHA does not pose a genotoxic risk and shows no carcinogenic potential in mammals lacking a forestomach.
Epidemiological evidence further indicates no increased cancer risk associated with typical dietary exposure to BHA [9]. Moreover, antioxidants such as BHA may exert beneficial effects by preventing the degradation of nutrients in foods and inhibiting the formation of toxic compounds resulting from fatty acid oxidation [10].
Based on the findings of multiple studies, the International Agency for Research on Cancer (IARC) has acknowledged that BHA shows no genotoxicity in bacterial or mammalian test systems. Furthermore, IARC has reported that when tested in combination with other chemicals—typically known mutagens or carcinogens—BHA reduced the activity of these mutagens/carcinogens [11].
In 2003, IARC convened a dedicated workshop, during which experts concluded that BHA does not pose a safety concern for humans [12].
BHA as an Antioxidant
As a food additive, the primary focus of research on BHA has been on repeat-dose toxicological studies and the establishment of safe intake levels. However, it is also important to recognize that several studies have reported potential health benefits associated with low-level BHA exposure.
Both endogenous and exogenous (dietary) antioxidants play an important role in human health by inhibiting the propagation of reactive oxygen species (ROS) and other free radicals that are continuously generated during normal physiological processes. These free radicals are implicated in the pathogenesis of many human diseases, including cancer.
Dietary antioxidants occur naturally in foods—such as flavonoids, arginine, vitamin C, vitamin E, carotenoids, resveratrol, and selenium—and are also commonly added to foods to suppress oxidative processes that lead to spoilage. Intentionally added antioxidants include both naturally occurring substances and synthetic antioxidants, such as BHA.
There is no scientific basis for concluding that the “natural” or “synthetic” origin of an antioxidant is inherently related to its safety. Notably, synthetic antioxidants are often incorporated into foods because they are specifically designed to be effective at very low concentrations.
Regulatory Standards
The safety of BHA as an antioxidant used in the food supply has been confirmed by regulatory authorities, including the U.S. Food and Drug Administration (FDA), the European Food Safety Authority (EFSA), Health Canada, and the Joint FAO/WHO Expert Committee on Food Additives (JECFA), as well as by the scientific research community [14]. Accordingly, maximum permitted levels (MPLs) for BHA have been established across a wide range of food categories.
United States FDA
FDA has conducted decades of research on the use of BHA in foods and has determined that, at current levels of use, BHA does not pose a safety concern. When the total content of antioxidants does not exceed 0.02% of the fat or oil content (including essential/volatile oils present in foods), BHA is generally recognized as safe (GRAS) [14].
For certain food categories—such as beverages and desserts prepared from dry mixes—the permitted level of BHA is limited to 2 ppm. For other foods, such as active dry yeast, the permitted level may be as high as 1,000 ppm [3]. The FDA also requires food manufacturers to declare BHA as an additive on product labels.
European Food Safety Authority (EFSA)
Under Directive 95/2/EC, BHA is authorized for use as a food additive (antioxidant), with maximum permitted levels specified for different food categories. The European Food Safety Authority (EFSA) conducted an updated dietary exposure assessment for BHA [4] and concluded that both average and high-level intakes among children and adults are unlikely to exceed the acceptable daily intake (ADI) of 1.0 mg/kg body weight per day. Therefore, no safety concerns were identified under current authorized uses.
Joint FAO/WHO Expert Committee on Food Additives (JECFA)
The Joint FAO/WHO Expert Committee on Food Additives (JECFA) first evaluated the safety of BHA as a food additive in 1961, followed by several re-evaluations during the 1980s. Based on the dose-dependent and reversible nature of lesions observed in rats, the Committee established an acceptable daily intake (ADI) for BHA of 0.5 mg/kg body weight per day [7].
Summary
As a synthetic antioxidant, BHA plays an important role in protecting food safety through its antioxidant properties. Numerous studies have demonstrated that BHA does not pose a safety concern to humans at levels typically found in the diet.
When used in compliance with regulatory standards in foods or animal feed, there is no cause for concern, as the amounts of BHA added are extremely low and well within established safety limits.
About L&P Food Ingredient Co., Ltd
L&P Food Ingredient Co., Ltd (L&P Foods) is a solution provider for nutrition, health, and food safety. Backed by the strong technical support of the Guangdong Food Industry Institute and the Guangye Research Institute of Biotechnology, and through nearly four decades of continuous innovation and development, the company has built strong R&D capabilities and advanced technological expertise, securing a leading position within the industry nationwide.
L&P Foods holds comprehensive production qualifications, including valid manufacturing licenses and a full range of internationally recognized management system certifications, including Quality Management (ISO 9001), Food Safety Management (ISO 22000), Religious Certifications (KOSHER and HALAL), Occupational Health and Safety (ISO 45001), Environmental Management (ISO 14001), as well as the Global Standard for Food Safety (BRC). With consistent and reliable product quality, a well-established after-sales service system, and the capability to provide customized solutions tailored to customer needs, L&P Foods has become a trusted and preferred supplier for downstream manufacturers.
Reference:
[1]https://www.who.int/zh/campaigns/world-food-safety-day/2022
[2] ILSI (International Life Sciences Institute)Butylated Hydroxyanisole (BHA). A Monograph(1984)Washington DC.
[3] 21CFR172.110
[4] EFSA J.,9(10)(2011),p.2392.
[5] Health Canada List of permitted preservatives (lists of permitted food additives)(2020)
[6] JECFA (Joint FAO/WHO Expert Committee on Food Additives)Evaluation of certain food additives and contaminants (Thirty-third Report of the joint FAO/WHO Expert committee on food additives)(1989).WHO Technical Report Series 776
[7] Environment Canada,Health Canada.Draft screening assessment for the challenge, phenol,(1,1-dimethylethyl)-4-methoxy-(butylated hydroxyanisole),chemical abstracts service registry number 25013-16-5.Existing Substances Evaluations,1-36(2010).
[8] Botterweck et al.,Fd Chem. Toxicol.,38(7)(2000),pp.599-605.
[9] Grice et al., Fd Chem. Toxicol.,24(1986),pp.1235-1242.
[10] IARC (International Agency for Research on Cancer)Butylated hydroxyanisole (BHA)Some Naturally Occurring and Synthetic Food Components, Furocoumarins and Ultraviolet Radiation. IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Humans,vol.40,International Agency for Research on Cancer,Lyon, France(1986),pp.123-159.
[11] IARC (International Agency for Research on Cancer)Predictive Value of Rodent Forestomach and Gastric Neuroendocrine Tumours in Evaluating Carcinogenic Risks to Humans.World Health Organization (WHO), International Agency for Research on Cancer (IARC),Lyon,France(2003).
[12] Susan P. et al..Regulatory Toxicology and Pharmacology.Volume 121, April 2021, 104887.
[13] Whysner and Williams.Pharmacol.Ther.,71(1/2)(1996), pp.137-151.
[14] 21CFR182.3169
[15] Davoli et al.,Rapid Commun.Mass Spectrom.,31(10)(2017), pp.859-864.
Latest Blogs
-
May. 07, 2026
【Brain Health Ingredient Series 7】Taurine – The “Natural Amino Acid Guardian” for Brain Protection
-
Apr. 27, 2026
-
Apr. 22, 2026
Exhibition Recap | L&P Foods Drives Quality & Efficiency at 2026 China Feed Industry Expo
-
Apr. 20, 2026
-
Apr. 17, 2026
【Brain Health Ingredient Series 4】 Citicoline (CDPC): The “Phospholipid Precursor” for Neural Repair




