Palmitoylethanolamide (PEA) as a Promising New Option for Migraine Relief
Feb. 21, 2024
Migraine is a common neurological disorder that primarily affects adolescents and middle-aged adults and is more prevalent in women. Globally, approximately 1.04 billion people suffer from migraine, with a lifetime prevalence of about 10% in men and 22% in women. Recurrent and long-term migraine attacks can lead to significant health impairment, reduced quality of life, and loss of productivity, making migraine one of the major global public health challenges.
Although a variety of pharmacological treatment options are available, many are associated with undesirable side effects. Emerging research suggests that palmitoylethanolamide (PEA) may represent a promising new option for alleviating migraine symptoms.

Image source: Pexels
On February 9, 2024, a recent double-blind, placebo-controlled clinical trial published in the journal Pharmaceuticals demonstrated that Gencor’s palmitoylethanolamide (PEA) product, Levagen+, can help alleviate discomfort associated with migraine. The study also found that Levagen+ reduced the duration of migraine episodes in otherwise healthy individuals.
The study enrolled 80 participants experiencing migraine symptoms. Participants were randomly assigned to two groups and received either 600 mg of Levagen+ or a maltodextrin-based placebo. Pain intensity was recorded using a Visual Analog Scale (VAS) every 30 minutes for up to 4 hours, or until the migraine resolved. If migraine symptoms persisted after 2 hours, participants were instructed to take a second dose.
The results showed that, at both the 2-hour and 8-hour time points, Levagen+ was more effective than placebo in relieving headache symptoms. In addition, compared with the placebo group, participants in the Levagen+ group reported lower VAS pain scores at 1.5 hours and 4 hours, and Levagen+ significantly reduced the need for additional medication use.
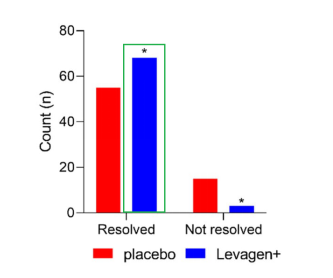
Figure 1. Number of Participants with Migraine Relief after 8 Hours
(Levagen+ group vs. placebo group: 68 vs. 55; p = 0.0022, * indicates a statistically significant difference compared with the placebo group.)
Image Source: Pharmaceuticals (2024), Briskey et al.
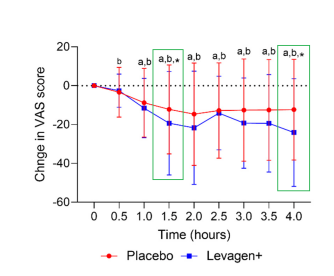
Figure 2. Changes in Visual Analog Scale (VAS) Pain Scores during the First 4 Hours
(*indicates p < 0.05, showing a statistically significant difference compared with the placebo group.)
Image Source: Pharmaceuticals (2024), Briskey et al.
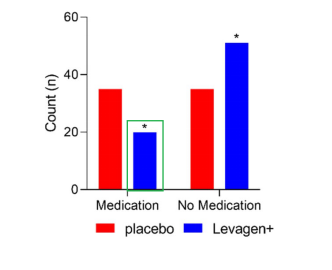
Figure 3. Number of Medications Used as Reported at 8 Hours
(*indicates p < 0.05, showing a statistically significant difference compared with the placebo group.)
Image Source: Pharmaceuticals (2024), Briskey et al.
PEA has a long history of use in a wide range of clinical conditions and has been shown in the scientific literature to exert analgesic, anti-inflammatory, immunomodulatory, and neuroprotective effects. PEA can be applied to support joint health, muscle recovery, sleep quality, allergy relief, and the alleviation of upper respiratory tract infection–related symptoms. It is considered a well-tolerated dietary supplement with no known significant side effects.
About L&P Food Ingredient Co., Ltd
L&P Food Ingredient Co., Ltd (L&P Foods) is a professional manufacturer specializing in food additives and functional food ingredients. The company holds comprehensive production qualifications, including valid manufacturing licenses and a full range of internationally recognized management system certifications, including Quality Management (ISO 9001), Food Safety Management (ISO 22000), Religious Certifications (KOSHER and HALAL), Occupational Health and Safety (ISO 45001), Environmental Management (ISO 14001), as well as the Global Standard for Food Safety (BRC). With consistent and reliable product quality, a well-established after-sales service system, and the capability to provide customized solutions tailored to customer needs, L&P Foods has become a trusted and preferred supplier for downstream manufacturers.
References:
Briskey, D.; Skinner, R.; Smith, C.; Rao, A. Effectiveness of Palmitoylethanolamide (Levagen+) Compared to a Placebo for Reducing Pain, Duration, and Medication Use during Migraines in Otherwise Healthy Participants—A Double-Blind Randomised Controlled Study. Pharmaceuticals 2024, 17, 145.
Latest Blogs
-
May. 07, 2026
【Brain Health Ingredient Series 7】Taurine – The “Natural Amino Acid Guardian” for Brain Protection
-
Apr. 27, 2026
-
Apr. 22, 2026
Exhibition Recap | L&P Foods Drives Quality & Efficiency at 2026 China Feed Industry Expo
-
Apr. 20, 2026
-
Apr. 17, 2026
【Brain Health Ingredient Series 4】 Citicoline (CDPC): The “Phospholipid Precursor” for Neural Repair




